Research
Autophagy, the cellular waste disposal system
We aim at discovering the roles and rules of autophagy: the process of cellular waste disposal. During autophagy, cytoplasmic material is enclosed within autophagosomes and transported to the lytic compartment (the vacuole in yeast, the lysosomes in mammals) for degradation and recycling. Autophagy is emerging as a central biological pathway that promotes health and longevity. On the other hand, defects in autophagy have been associated with several human diseases, including neurodegenerative disorders and cancer. Our goal is to dissect autophagy at a molecular level, so that we can understand its dysfunction in disease and unleash its potential as a therapeutic target.
The core autophagic machinery is highly conserved from yeast to humans. Therefore, we use both mammalian cell culture and budding yeast as model systems to discover the basic principles of autophagy. Our approaches range from classical reconstitutions, using recombinant proteins and purified membranes, to reconstitutions using cell fractions and synthetic in vivo approaches. We also analyze intact cells.
Taken together, we aim at recreating the full complexity and functionality of autophagy in vitro, to then understand the process in vivo. We strongly believe that curiosity-driven basic research is the key to innovation, as many scientific breakthroughs were based on unanticipated outcomes of basic research. Our goal of understanding the fundamental molecular mechanisms of autophagy will shed light on how autophagy functions in more complex situations, such as in organisms and diseases. Basic research will thus help to bring autophagy to clinics and pharmacies, likely by yet unforeseeable routes.
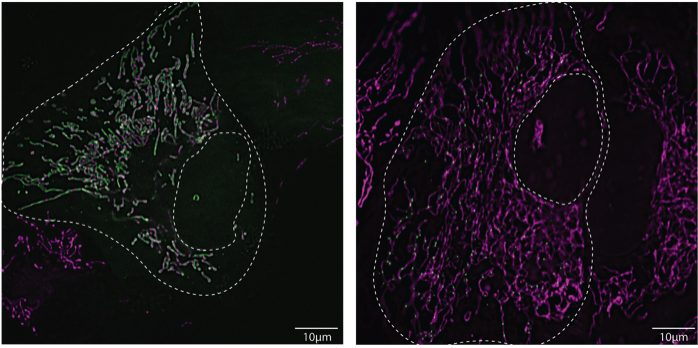
Figure: Differential assembly of the autophagy machinery (green) during mitophagy on mitochondria (magenta). U2OS cells, live cell imaging